Endothermic & Exothermic Chemical Reactions
Endo & Exo-thermic reactions
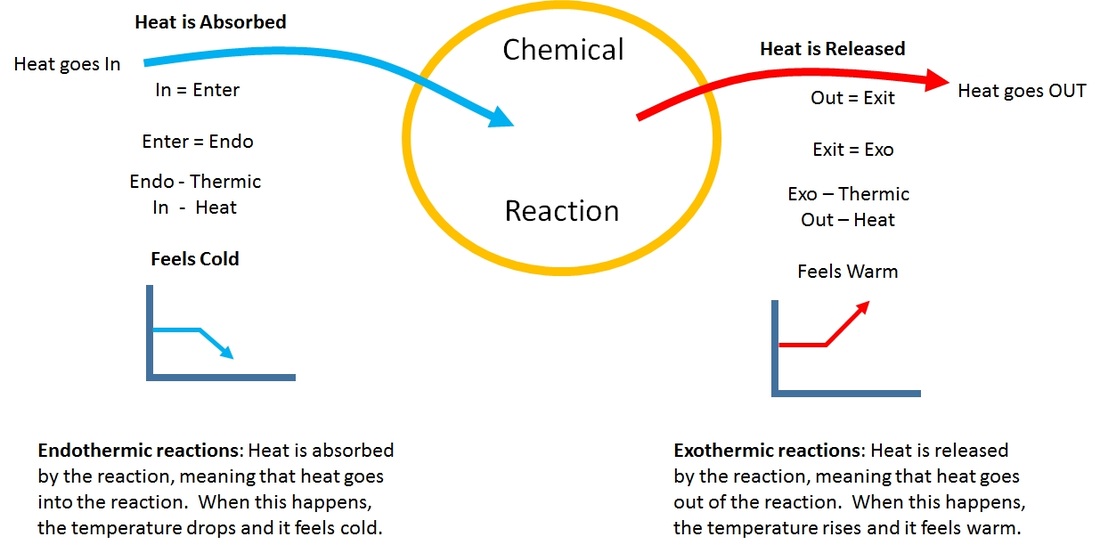
Endo and Exothermic reactions are always in the perspective OF the reaction - NOT BY YOU OR THE THERMOMETER!
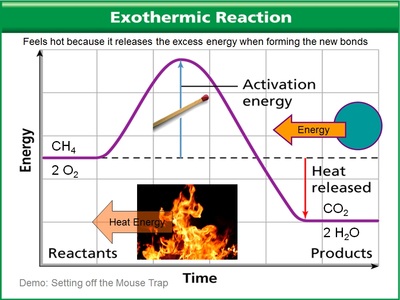
Exo (outside) thermic (heat). Heat goes out of the reaction. In other words, heat is RELEASED BY the reaction. Meaning, it feels warm to you and me.
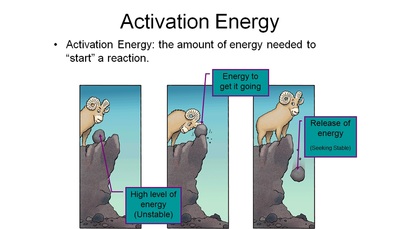
Exothermic reactions are like a mousetrap going off. Once the trap is tripped, the stored energy in the spring is released - unleashing an enormous amount of energy.
Endo and Exothermic reactions are always in the perspective OF the reaction - NOT BY YOU OR THE THERMOMETER!
Exo (outside) thermic (heat). Heat goes out of the reaction. In other words, heat is RELEASED BY the reaction. Meaning, it feels warm to you and me.
Exothermic reactions are like a mousetrap going off. Once the trap is tripped, the stored energy in the spring is released - unleashing an enormous amount of energy.
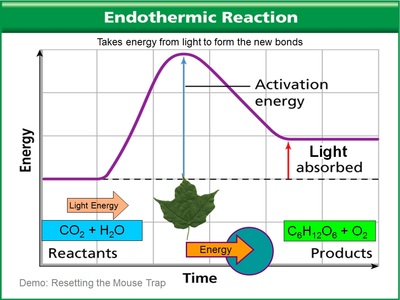
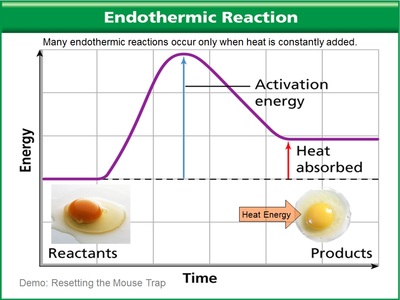
Endo (inside) thermic (heat). Heat goes into the reaction. In other words, heat is ABSORBED BY the reaction. What this translates into is that you feel cold because the heat is taken from you to go into the reaction.
Endothermic reactions are like setting the mousetrap - in that it requires an outside source of energy to reset the trap, much like it takes heat energy to make chemicals come together in this reaction. Photosynthesis is much like this... the plant absorbs light energy instead of heat energy to react carbon dioxide and water to form higher levels of chemically reactive chemicals such as sugar and oxygen.
Endothermic reactions are like setting the mousetrap - in that it requires an outside source of energy to reset the trap, much like it takes heat energy to make chemicals come together in this reaction. Photosynthesis is much like this... the plant absorbs light energy instead of heat energy to react carbon dioxide and water to form higher levels of chemically reactive chemicals such as sugar and oxygen.
Lab: Which reaction is endothermic?
In this lab, we asked the question: "Which reaction is endothermic?"
More specifically, it was between these two reactions:
(One other reaction can include: Ammonium Nitrate & Water)
In this case, the hypothesis is stated slightly differently - based on your background knowledge. It may start out as simple as "I hypothesize that" or "I believe that the (insert your reaction choice here) because (base it on basic background knowledge).
Materials:
Basic Procedures
Students are to incorporate the following in their procedures:
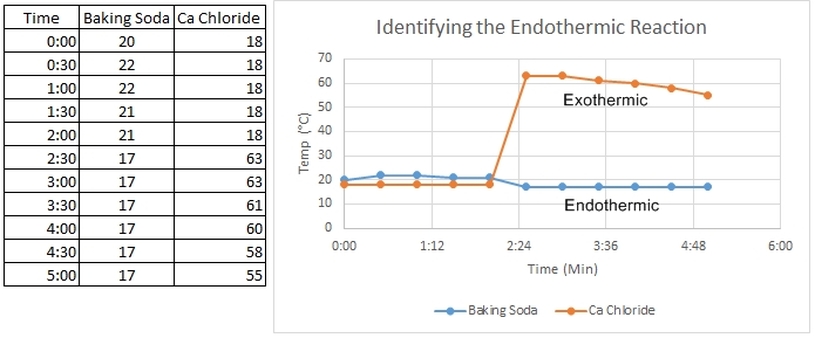
Below is the data of a group of students.
Form your own set of data analysis statements and conclusion.
More specifically, it was between these two reactions:
- Calcium Chloride & Water (Calcium Chloride is used to melt ice on your driveway)
- Baking Soda & Citric Acid (Baking soda is heavily used in many baking products. Citric Acid is found in Lemon Aide and all citrus fruit).
(One other reaction can include: Ammonium Nitrate & Water)
In this case, the hypothesis is stated slightly differently - based on your background knowledge. It may start out as simple as "I hypothesize that" or "I believe that the (insert your reaction choice here) because (base it on basic background knowledge).
Materials:
- 10g Calcium Chloride
- 10g Baking Soda
- 20mL water
- 20mL Citric Acid solution
- Timer
- Thermometer
Basic Procedures
Students are to incorporate the following in their procedures:
- Measure the temperature in 30 second intervals
- Measure the temperature of the liquid at the base of the graduated cylinder
- Measure the temperature of the liquid for 2 minutes without the powder reactants (baking soda or Calcium Chloride)
- Measure the temperature of the liquid for 3 minutes with the powder reactants included.
Below is the data of a group of students.
Form your own set of data analysis statements and conclusion.